
Researchers claim to have solved an old problem: lithium batteries bursting into flames. This means it will be possible again to use batteries with a larger capacity than the current ion models, following a period when lithium batteries were out of favour because of the fire hazard they posed.
The ubiquitous lithium-ion battery delivers a lot of power, but is limited by its design: lithium ions are embedded in carbon, so the capacity per gram of battery is relatively low. Pure lithium is much more advantageous, something which has been known since the 1980s when the first liquid lithium batteries came on the market.
ACCUMULATION
However, it wasn’t all roses: these batteries, used for instance in the first mobile phones, regularly burst into flames when they were charged. It turned out that this was caused by dendrites – basically whiskers of lithium that grow inside batteries. These then accumulate too much energy in one place, overheating the battery.
Researchers at the University of Michigan solved this by applying a thin layer of ceramic to the lithium in the battery. The ceramic works as an electrolyte, the medium between the two ends of the battery. The ceramic prevents lithium dendrites from forming, and results in a battery which combines safety with all the advantages of a pure lithium battery. In theory, such a battery has a capacity ten times that of a comparable lithium-ion battery.
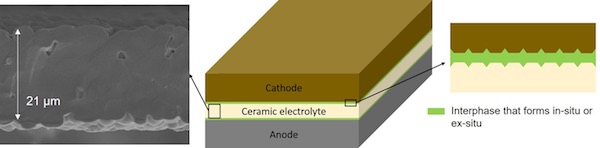
SMOOTH LAYER
This trick has been tried before, but was unsuccessful because the lithium grew through imperfections in the ceramic layer. The researchers at the University of Michigan solved this by simply making a pristine layer, which is now possible thanks to modern depositing technology. In the past, doing so with the equipment available was complicated. If the layer is completely smooth, there are no points where lithium can accumulate.
MORE RANGE FOR YOUR CAR
For the time being, this battery can only be made in the laboratory. However, if the ceramic layer is perfected, this type of battery could prove to be an enormous boost for electric cars, as well as other applications. Currently, electric cars need huge lithium-ion battery packs to have sufficient range. A liquid battery can store much more energy in a small surface, offering a choice of lighter cars or bigger ranges.
Image: Evan Dougherty/Michigan Engineering Communications & Marketing
If you found this article interesting, subscribe for free to our weekly newsletter!
Nieuwsbrief
Vond je dit een interessant artikel, abonneer je dan gratis op onze wekelijkse nieuwsbrief.

